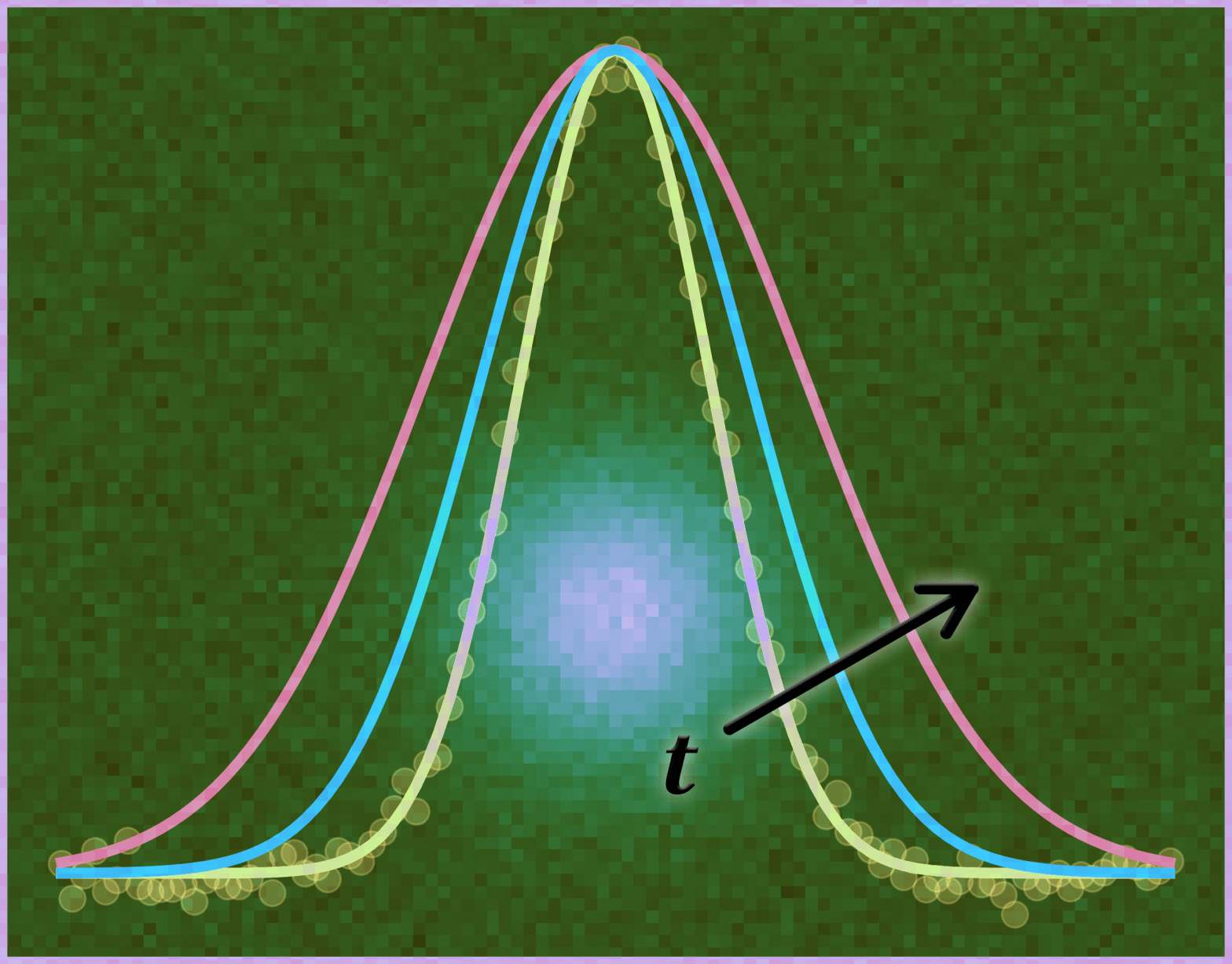
I am happy to announce this web application, designed to assist researchers working in pump-probe and time-resolved photoluminescence microscopy. This tool aims to make estimating the Contrast-to-Noise Ratio (CNR) for noisy 1-dimensional Gaussian profiles as simple and efficient as possible. It’s completely free to use. Contrast-to-Noise Estimator
Category Archives: Science
Is There Any Escape From Noise?
Exciting news! Our latest research, now an Editor’s Pick in The Journal of Chemical Physics, unveils how even moderate experimental noise can dramatically skew the accuracy of measuring excited state diffusion in materials like perovskites. Delve into our findings and explore our new open-source tools designed to refine these measurements. Join us in advancing the precision of diffusion measurements.
The multilayered role of scientific posters
A simple question from a 9-year-old at a major scientific conference sparks a reflection on the significance of scientific posters. Delve into how these visual aids cater to diverse cognitive preferences, making research findings more accessible and inclusive.
The etymology and meaning of methyl, ethyl, propyl, butyl
Chemistry study often feels like learning a new language due to its extensive vocabulary. Understanding word origins can greatly improve memory retention. Early chemistry nomenclature includes memorizing roots like methyl, ethyl, propyl, and butyl, representing 1 to 4 carbon chains in functional groups. This article offers a thorough explanation for those studying or interested inContinue reading “The etymology and meaning of methyl, ethyl, propyl, butyl”
Penrose Tessellation Cookie Cutters
Over the holiday break, I took a little time for a side quest that engaged my creativity while involving mathematics, 3D modeling, and baking. I designed a pattern based on Penrose tiles and made it into cookies.
Double Chocolate Pecan Tau Pie
When you want to celebrate the true fundamental circle constant on Tau Day (June 28), there is no better way than with a double chocolate pecan tau pie.
The Etymology and Meaning of Anode and Cathode
The terms “anode” and “cathode” were first published by Michael Faraday, F.R.S. in 1834. A delightful (and highly recommended) historical account of how these words were conceived by Faraday and his associates can be found in Faraday Consults the Scholars: The Origins of the Terms of Electrochemistry by Sydney Ross [1]. As scientists have learnedContinue reading “The Etymology and Meaning of Anode and Cathode”
Schrödinger’s Cat, a sonnet
Should quantum physics e’er be standardized, when taken in a thought experiment, its terms of meaning judged and analyzed, absurdity prevails, not merriment. A cat both dead and living cannot be. That was the point old Erwin tried to make. To measure is to interfere, you see, some photon must be thrown to cause aContinue reading “Schrödinger’s Cat, a sonnet”
Tau Pendant Video
Take a look at this new video promoting the Tau open circle pendant I designed.
China Coatings Journal
I’m a co-author of the following article, published in two parts in the China Coatings Journal: